Article about the Equation of loupe magnifying glass
As explained in the topic loupe (magnifying glass), an object appears small when viewed from a great distance and looks great when viewed at close range. The difference in the size of the object seen by the eye is caused by the difference in angle between the eye and the object. When an object is very far away from the eye, the angle between the eye and the object is smaller so that the image formed on the retina of the eye is also smaller. Conversely, when the object is close to the eye, the angle between the eye and the object is larger so that the image formed on the retina of the eye is also more significant. The closer to the eye, the higher the angle between the eye and the object, so that the image that forms on the retina is also getting bigger. Kindly note that the average human eye has a near point is 25 cm so that the distance between the eye and the object cannot be smaller than 25 cm. It can be concluded that the angle between the normal eye of an average human and an object is maximum when the distance between the eye and the object is 25 cm.
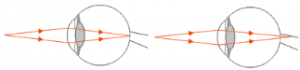 Based on the previous explanation, it can be concluded that the cause of farsightedness is the cornea and the lens of the eye is less curved,
Based on the previous explanation, it can be concluded that the cause of farsightedness is the cornea and the lens of the eye is less curved,